Research
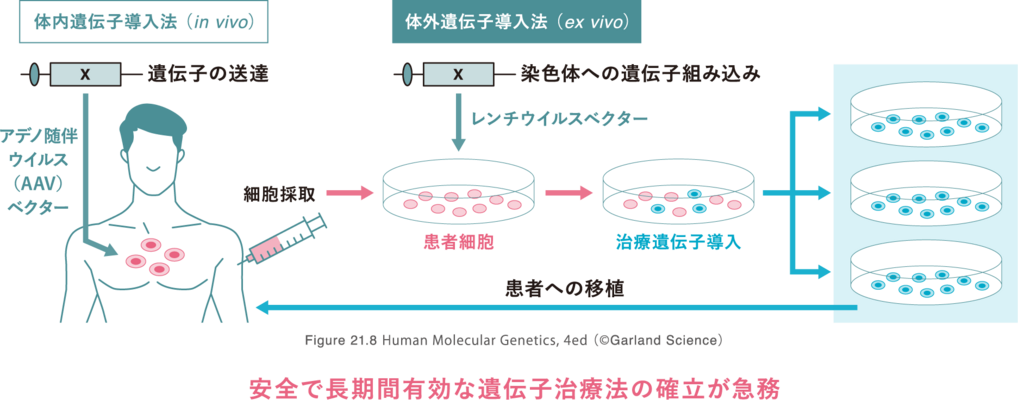
Development of Viral Vector Related Technologies
We are now developing novel variants of adeno-associated virus (AAV) vectors, that are expected to have high degree of safety and be capable of expression over longer periods of time, as well as basic technologies for their production and purification in order to achieve full-scale commercialization as gene therapy products.
We have also developed host cells with enhanced efficacy for vector production, and improved cell culture methods using bioreactors as well as the analysis of viral particles secreted into the medium are also under investigation. Regarding purification methods, in addition to the development of new ultracentrifugation technologies we are also promoting the creation of processes that combine ultrafiltration, ammonium sulfate precipitation, ion exchange, gel filtration and hydrophobic chromatography according to different purposes. To produce high-quality viral vectors that have minimal contamination with empty and incomplete particles, we are focused on the development of next-generation analytical techniques reflecting the guidelines of the FDA and domestic regulatory institutions, including the use of analytical ultracentrifugation and cryo-electron microscopy.
In addition, as a technology related to AAV vectors, we are also developing a non-viral DDS using hollow particles without the viral genome. While keeping the functional analysis of novel capsid mutants, we are also developing technologies to encapsulate or bind plasmids, DNA fragments or artificial nucleic acids to the empty particles and propose their application as nucleic acid therapeutics.